sábado, 28 de mayo de 2011
Demencia autoinmune
 |
Neuroimaging in patients with an immunotherapy-responsive cognitive disorder.
Magnetic resonance imaging: yellow arrows indicate areas of abnormality on fluid-attenuated inversion recovery (FLAIR). A, 36-year-old woman (patient 10 in Appendix 1) had fluctuating memory problems and was seropositive for glutamic acid decarboxylase-65 autoantibody. Bilateral hippocampal axial FLAIR abnormality, shown in A1, almost completely resolved after treatment with intravenous (IV) methylprednisolone (A2). B, 51-year-old woman (patient 20 in Appendix 1) had subacute fluctuating memory problems, multifocal neurologic examination findings, and evidence of autoimmunity (IgM antiphospholipid antibody). Symmetric confluent T2 signal abnormality in the white matter of both hemispheres (B1) decreased after treatment with IV methylprednisolone (B2). C, 60-year-old man (patient 41 in Appendix 1) had memory, language, and gait problems and was seropositive for both striational and glutamic acid decarboxylase-65 antibodies. Axial T1 magnetic resonance imaging with contrast demonstrated periventricular vessel enhancement (C1) and resolution after treatment (C2). D, 53-year-old woman (patient 29 in Appendix 1) had memory loss, hallucinations, and subsequent seizure; cerebrospinal fluid protein was elevated (>100 mg/dL), and she was seropositive for thyroid peroxidase antibodies and neuronal and muscle acetylcholine receptor antibodies. Axial FLAIR images show diffusely increased T2 signal in the midbrain (D1), which improved after treatment with IV methylprednisolone (D2). Multiple myeloma was diagnosed 18 months after neurologic presentation.
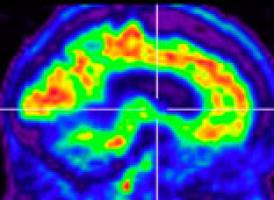
Positron emission tomographic imaging: Brain reconstructions (brighter color represents regions of hypometabolism) in a 58-year-old man (patient 21 in Appendix 1) who presented with personality change and memory problems and had elevated cerebrospinal fluid protein (>100 mg/dL). Hypometabolism, predominantly frontal and temporal (E1), improved after treatment with IV methylprednisolone (E2).
Single-photon emission computed tomographic brain imaging: Brain neuroimaging in a 35-year-old man (patient 22 in Appendix 1) who presented with vertigo and memory problems, had multiple coexisting autoimmune conditions, and was seropositive for muscle acetylcholine receptor and striational antibodies. Diffuse decrease in uptake in frontotemporoparietal regions (F1) was markedly improved globally after treatment with IV methylprednisolone (F2).
|
Encefalitis antiNMDA simulando neuromielitis óptica seronegativa
Resonancia de alto campo del hipocampo
viernes, 13 de mayo de 2011
Uso del PET con Florbetapir para la detección del beta amiloide en la Enfermedad de Alzheimer
El diagnóstico precoz de los pacientes con enfermedad de Alzheimer (EA) supone un constante desafío.Se estima que entre un 10-20% de los pacientes diagnosticados de EA en realidad no la presentan en el estudio anatomopatológico y que un 33% de pacientes con síntomas leves no pueden ser diagnosticados de EA. Por ello, los marcadores biológicos se han propuesto como una solución a este problema.
Recientemente ha sido publicado un estudio, todavía preliminar, (JAMA. 2011;305(3):275-283) de un nuevo biomarcador (Florbetapir-F 18) para su uso en imágenes por Tomografía por emisión de positrones (PET). Los autores demuestran en el estudio una correlación de las imágenes de Florbetapir-PET "in vivo" con la densidad de beta-amiloide en los estudios necrópsicos postmorten.
El Florbetapir se une al Compuesto B Pittsburgh como marcador para imágenes de PET que muestra la presencia "in vivo" de los depósitos cerebrales de beta-amiloide propios de la EA. La ventaja del Florbetapir-F 18 con respecto al Compuesto B Pittsburgh, es que este último al usar el Carbono-11 como isótopo presenta una menor vida media (20 minutos) que el Fluor-18 (isótopo del Florbetapir).
Los estudios previos demostraban la alta afinidad y especificidad del Florbetapir con el Beta-amiloide. Además, su rápido aclaramiento (en el caso de que no se haya fijado al beta-amiloide) cerebral lo hace ser un excelente biomarcador para su uso en el PET.
jueves, 12 de mayo de 2011
sábado, 7 de mayo de 2011
Embolismo gaseoso
Paciente con derrame pleural metaneumónico que tras tratamiento con pleurodesis quimica con fibrinolíticos sufre embolismo cerebral gaseoso. Recuperación completa tras tratamiento en cámara hiperbárica.
(Texto completo)
viernes, 1 de abril de 2011
viernes, 18 de marzo de 2011
Secuencias de difusión en RM para el estudio del mecanismo de deglución
Introduction Understanding the relevance of neural pathways that control human swallowing and related behaviours is paramount to correctly identifying and treating swallowing disorders. We aimed to evaluate these connections using complimentary imaging modalities in vivo.
Methods In 13 healthy adults (3 Male/10 Female, 27–62 years old) Transcranial Magnetic Stimulation (TMS) evoked pharyngeal motor responses were recorded via a swallowed intraluminal catheter and used to assess motor cortical pharyngeal representation in both hemispheres.1 Thereafter, event-related 3T functional magnetic resonance imaging (fMRI) was applied to assess blood-oxygen-level-dependent signal changes (compared to rest) in response to a pseudo-randomised order of water and saliva swallowing in addition to a tongue elevation task. Swallow related behaviour was monitored using a pressure feedback belt around the subjects' neck. Distortion-corrected diffusion weighted imaging (DWI) data were then acquired for probabilistic tractography to determine anatomical fibre tracts. Task-related fMRI activations were identified in SPM8 and used to seed unconstrained multi-fibre probabilistic tractography independently in each hemisphere.2 These results were compared with TMS inter-hemispheric differences.
Results TMS data revealed 12/13 subjects had asymmetric motor representation (lateralised dominance) for the pharynx (6 left, 6 right) with significantly larger responses observed in the lateralised projection (p < 0.001). FMRI data (figure 1) also revealed asymmetric activations in both sensorimotor cortices in each subject for the different tasks which were used as seed regions for tractography. Largest activations were observed in primary motor cortices (Brodmann Area (BA) 4) after water swallowing, predominantly right lateralised. By contrast, tongue elevation and saliva swallowing produced a greater number of left lateralised brain areas, particularly pre- and supplementary-motor cortex (BA 6) as well as areas involved with language and speech processing (BA 22,40,44). Tractography revealed larger tract volumes in the hemispheric dominant projection predicted by TMS compared to the non-dominant projection for water but not for saliva swallowing or tongue elevations (p < 0.001).
Figure 1
OC-078 fMRI activation for swallowing and related behaviours
Conclusion Using multimodal imaging we have performed a detailed mapping of the network of neural pathways that contribute to human swallowing and its related behaviours. Our data also provide further evidence for cerebral asymmetry and its relevance in the control of human swallowing.
Suscribirse a:
Entradas (Atom)